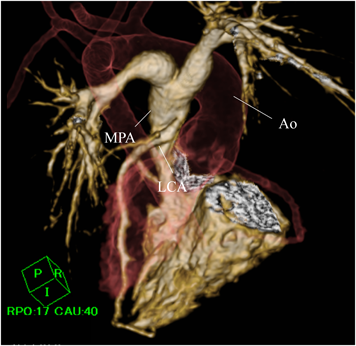
A female neonate weighing 3,148 g was born by induced labor at 39 weeks of gestation. The patient was prenatally diagnosed with TOF at 28 weeks of gestation. After birth, her systemic oxygen saturation was 95%. She exhibited tachycardia during feeding and edema. Electrocardiography (ECG) showed ST-segment depression in II, III, aVF, and V3−V6, with a heart rate of 168 beats per minute. She was transferred to our hospital at 20 days of age. Echocardiography revealed that the aorta was overriding the ventricular septum with a doubly committed juxta-arterial ventricular septal defect (VSD) and a hypoplastic pulmonary valve, suggesting TOF. Her left ventricular ejection fraction (LVEF) had decreased to 48%. Additionally, Doppler echocardiography showed lack of continuity between the left coronary artery (LCA) and the ascending aorta with abnormal blood flow draining into the main pulmonary trunk and mild mitral regurgitation, suggesting ALCAPA. Contrast-enhanced computed tomography (CT) was performed to determine the spatial relationship between the LCA and the main pulmonary trunk. Volume-rendered images revealed that the LCA originated from the posterior sinus of the main pulmonary trunk (Fig. 1). Cardiac catheterization revealed that the mean pulmonary arterial pressure and ratio of pulmonary to systemic flow were 15 mmHg and 4.1, respectively. Right ventriculography showed a pulmonary annulus diameter of 6 mm (Z-score: −2.71). Aortography and pulmonary arteriography revealed that the LCA was retrogradely opacified from the right coronary artery (RCA) through the inter-coronary collaterals. We decided to plan a staged repair; reimplantation of the LCA followed by intracardiac repair of TOF.
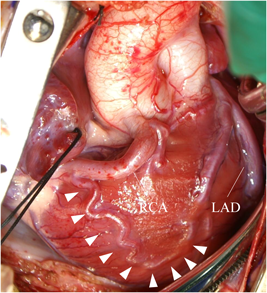
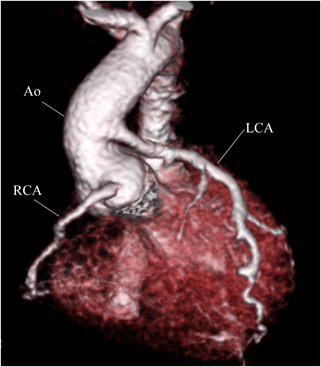
At 2 months of age, the infant weighed 3,700 g. We performed direct LCA implantation onto the ascending aorta. Intraoperatively, the RCA was found dilated and tortuous as its collateral circulation had flowed toward the left anterior descending artery (Fig. 2). After establishing cardiopulmonary bypass (CPB), a cardioplegic catheter was placed in the ascending aorta. During the first cardioplegic infusion through the ascending aorta after aortic cross-clamping, the left and right pulmonary arteries were snared to prevent a flow of the cardioplegic solution into the peripheral pulmonary artery (PA). The main PA was transected transversely distal to the LCA orifice, and the LCA ostium was excised as a button with a generous cuff of the main PA wall. The LCA button was mobilized to facilitate anastomosis to the ascending aorta, avoiding tension or kinking. In addition, selective infusion of cardioplegic solution was carried out into the LCA button using a 6 Fr injection tube during the implantation maneuver. The LCA button was anastomosed to the left side of the ascending aorta using a running suture. The defect created at the main PA sinus was repaired with an autologous pericardial patch, and the main PA was reconstructed by anastomosis to the distal PA. Weaning of the patient from CPB was uneventful, and the sternum was closed. Although she required peritoneal dialysis for 4 days, the patient was extubated on the 5th postoperative day. Subsequent recovery was uneventful. LVEF was maintained at 62% with mild mitral regurgitation. Postoperative ECG showed that ST segment depression, which had been detected during preoperative tachycardia, was no longer observed. The CT on the 11th day following ALCAPA repair revealed no obstruction across the transplanted LCA arising from the ascending aorta (Fig. 3).
At 12 months of age, cardiac catheterization prior to intracardiac repair showed high pulmonary blood flow with a pulmonary to systemic flow ratio of 1.6 in the setting of TOF, mild hypoplastic pulmonary annulus (8.5 mm-diameter, Z-score, −2.21), LVEF 65%, and no mitral regurgitation. Additionally, blood flow through the transplanted LCA was smooth. The patient underwent intracardiac repair of TOF when she weighed 8,300 g. Intraoperatively, the pulmonary valve was found to be longitudinally bicuspid, with a diameter of 8 mm. The doubly committed juxta-arterial VSD was large and extended to the pulmonary valve annulus. To close the large VSD, we placed two sets of 6-0 monofilament sutures with 3 mm spaghetti through either the pulmonary valve annulus or the tricuspid valve. Subsequently, we reconstructed the right ventricular outflow tract (RVOT) using a transannular patch because a large VSD baffle was present just below the pulmonary valve; the transannular patch bearing a monocuspid fan-shaped polytetrafluoroethylene valve. The postoperative course was uneventful. Cardiac catheterization on the 16th postoperative day showed that the right ventricular systolic pressure was as low as 24 mmHg, and blood flow through the transplanted LCA was smooth. Moreover, pulmonary regurgitation was less than mild. The patient was discharged on the postoperative day 21. She was in a good condition at 4 years of age in the outpatient clinic follow-up.
Coronary arterial abnormality occasionally occur in 4–6% of patients with TOF; however, ALCAPA is rarely associated with TOF.2) With these lesions in combination, ischemic changes would depend on the inter-coronary collaterals, coronary arterial dominance, and severity of pulmonary stenosis.3) Compared with patients solely with ALCAPA, those with both ALCAPA and TOF may demonstrate myocardial ischemia earlier, because pulmonary arterial pressure would be lower and LCA flow from the pulmonary artery should be reduced.
In several reports, patients aged 19 months to 9 years who were diagnosed with ALCAPA and TOF were treated in one-stage radical surgery.1, 3–7) Only Bitar et al. reported an infant with ALCAPA and TOF undergoing one-stage repair.8) In their case, dehiscence of the VSD patch was repaired 2 weeks after the initial surgery when the patient was 2 months old. In another article, in contrast, 2 patients undergoing a staged approach died after the initial procedure. One of these patients had scimitar syndrome and right lung hypoplasia, and underwent LCA implantation together with construction of a central shunt. The patient died due to respiratory failure. In the other patient presenting low output syndrome, ALCAPA remained undiagnosed, and a Blalock-Taussig shunt was constructed. The patient died, and postmortem autopsy revealed ALCAPA.5)
In our present case, a clear sign of myocardial ischemic was detected on ECG, and led to a correct diagnosis soon after birth. The risk of anoxic attack was low owing to the large doubly committed juxta-arterial VSD with mild RVOT stenosis. Given the challenging nature of closing the large VSD in a 2-month-old infant weighing 3.7 kg, we prioritized the ALCAPA repair at that age. Although peritoneal dialysis was required perioperatively due to decreased left ventricular function immediately after the ALCAPA repair, her cardiac function gradually improved. This led to a stable intracardiac repair of TOF eventually. We believe that the staged repair management for this unusual combination of ALCAPA and TOF was safely and successfully achieved.
Author Contribution
Conceptualization: Yusuke Nakata, Yoshie Ochiai, Writing-original draft: Yusuke Nakata, Writing-review & editing: Yoshie Ochiai, Jun Muneuchi, Kunihiko Joo, Yuichirou Sugitani, Kohji Okamoto, Yoshiyuki Yamashita, Shigehiko Tokunaga, and Mamie Watanabe.