Chronic pulmonary regurgitation after repair of tetralogy of Fallot (TOF) may lead to right ventricular (RV) dilatation, which could be accompanied by ventricular tachycardia and sudden death.1–7) Several reports confirm that late pulmonary valve replacement (PVR) was a risk factor for postoperative mortality or morbidity.4, 6, 7) Most of patients with TOF who have undergone repair using a transannular patch (TAP) remain asymptomatic and free from medication during childhood. Accordingly, follow-up by cardiology specialists is easily interrupted in patients who live in remote areas. Since they have not been followed up by a cardiologist, there is a high probability that advanced right heart failure makes surgery infeasible, or that right heart function would not improve even if surgery is carried out. In fact, it is not clear how often postoperative TOF patients are lost to follow-up and how significant the impact of follow-up interruption would be. Therefore, we believe it is important to compare the group of patients who saw a cardiologist regularly after repair of TOF with the other group of patients who did not. The aim of this study is to reveal whether interruption of cardiologist follow-up affects PVR outcomes in patients with TOF who have previously undergone TAP repair.
Between 2007 and 2022, surgical PVR was performed in 15 patients with repaired TOF using a TAP at our institute. Symptoms before PVR were incidentally confirmed as hypoxemia in 1 patient, dyspnea in 5, and palpitation and arrhythmia in 1. The remaining 8 patients required therapeutic intervention due to exacerbation of catheter test results and echo findings. Then patients were divided into 2 groups by without (Group 1, n=4) or with (Group 2, n=11) scheduled follow-up by a cardiology specialist. Pre-operative characteristics and peri- and post-operative outcomes were retrospectively compared. The application of PVR was determined according to a previous report.8) Namely, significant pulmonary regurgitation plus any two or more findings from (2) to (8) are to be satisfied.
- (1) Pulmonary regurgitation moderate or severer on echocardiography
- (2) Pulmonary stenosis pressure gradient≧50 mmHg
- (3) Symptomatic patients
- (4) RV end-diastolic volume index≧160 mL/m2
- (5) Appearance of arrythmia
- (6) RV ejection fraction≦45%
- (7) A significant residual shunt present
- (8) Moderate or severe tricuspid regurgitation
PVR was performed using bioprosthetic valves in all cases. Implanted bioprostheses were Mosaic Ultra (Medtronic, Minneapolis, Minnesota) in 3 patients (before 2009), the Perimount Magna (Edwards Lifesciences, Irvine, CA) in 5 patients (after 2010), and INSPIRIS RESILIA (Edwards Lifesciences, Irvine, CA) in 7 patients (after 2019). The sizes of implanted valve prosthesis were 23 mm in 1patient, 25 mm in 11, and 27 mm in 3.
Study Methods
The present study is a retrospective, single institutional cohort study approved by the Saitama Medical University International Medical Center Institutional Review Board (Institutional approved number: 2023-077, 12/10/2023). The opt out consent was obtained instead of individual written informed consent because of retrospective nature of this study.
Evaluated variables were as follows: (1) differences in groups’ backgrounds, (2) Comparison of peri- and post-operative factors; the number of concomitant procedures, operation time, cardiopulmonary bypass time, amount of intraoperative bleeding, postoperative cardiothoracic ratio, postoperative serum brain natriuretic peptide, RV function indices assessed by magnetic resonance imaging including ejection fraction, end-systolic volume and its index, end-diastolic volume and its index, RV cardiac output, as well as pulmonary regurgitant fraction. The timing of each postoperative examination is the date of the latest postoperative visit in survivors and the date just before death in non-survivors.
Pre- and post-operative data were expressed by n (%) or median [interquartile range].
Statistical processing was performed using statistical software JMP Pro Ver.16 (Windows). A continuous variable that does not follow a normal distribution was compared using Mann–Whitney U test. A significant difference was defined as a risk rate of less than 5%.
Differences in Backgrounds of the Two Groups
Patient characteristics were summarized in Table 1. A median age at PVR was 49.0 years [43.0–53.2] in Group 1 and 23.0 years [21.5–38.0] in Group 2 (p=0.004). A median interval between TOF repair and PVR was 41.0 years [34.5–48.3] in Group 1 and 20.0 years [19.4–34.5] in Group 2 (p=0.004). Preoperative median cardio-thoracic ratio on chest-X-ray was 63% [57–86] in Group 1 and 55% [47–70] in Group 2 (p=0.022). Preoperative median concentration of brain natriuretic peptide was 100.5 pg/mL [57.6–131.9] in Group 1 and 24.7 pg/mL [17.5–28.9] in Group 2 (p=0.022). The preoperative sample for brain natriuretic peptide was collected within 3 months before surgery, and preoperative chest X-ray was taken within one week prior to surgery. No patients in Group 1 experienced surgical or catheter-based interventions before and after TOF repair. In contrast, those in Group 2 experienced various types of interventions such as construction of a Blalock-Taussig shunt, pulmonary artery stenting, right ventricular outflow tract (RVOT) ballooning etc. In Group 1, all patients underwent concomitant surgery in addition to PVR. Tricuspid annuloplasty was performed in 3 out of the 4 patients. Other concomitant procedures included mitral valve replacement, repair of partial anomalous pulmonary venous return, closure of coronary arterial fistula and RVOT reconstruction in this group (Table 2). In Group 2, PVR with or without RVOT reconstruction was carried out in 7 of 11 patients. In the other 4 patients, aortic valve replacement, tricuspid annuloplasty, or closure of left ventricular-right atrial shunt was performed. The Table 2 summarizes surgical procedures concurrently employed.
Table 1 Patient characteristics | Group 1 | Group 2 | p |
|---|
| Number of patients (n) | 4 | 11 | |
| Male patients (n) | 2 (50%) | 7 (64%) | 0.331 |
| Age at TOF repair (y) | 5.0 [3.5–7.0] | 3.0 [2.0–3.5] | 0.182 |
| Age at PVR (y) | 49.0 [43.0–53.2] | 23.0 [21.5–38.0] | 0.004 |
| Weight at PVR (kg) | 70.6 [56.9–82.9] | 53.1 [45.9–65.8] | 0.058 |
| TAP to PVR period (y) | 41.0 [34.5–48.3] | 20.0 [19.4–34.5] | 0.004 |
| Post-PVR follow-up period (y) | 1.1 [0.8–1.3] | 3.1 [2.8–11.0] | 0.005 |
| Pre-operative cardio-thoracic ratio (%) | 63 [57–86] | 55 [47–70] | 0.022 |
| Pre-operative BNP (pg/mL) | 100.5 [57.6–131.9] | 24.7 [17.5–28.9] | 0.022 |
| Pre-operativepulmonary regurgitant fraction (%) | 64.5 [46.8–82.3] | 58.6 [50.8–67.0] | 0.329 |
| Symptoms (dyspnea, hypoxia, palpitation) | 4 (100%) | 3 (27%) | 0.0002 |
| Valve regurgitation other than pulmonary artery valve more than mild-moderate (AR, MR, TR) | 3 (75%) | 8 (73%) | 0.468 |
| Data in the table are expressed as n (%) or median [interquartile range]. AR, aortic valve regurgitation; BNP, brain natriuretic peptide; MR, mitral valve regurgitation; PVR, pulmonary valve replacement; TAP, transannular patch; TOF, tetralogy of Fallot; TR, tricuspid valve regurgitation |
Table 2 Details of concomitant surgery in each group | Group 1 | Group 2 |
|---|
| Number of patients (n) | 4 | 11 |
| PVR | 0 | 3 |
| PVR+RVOTR | 0 | 4 |
| PVR+AVR | 0 | 1 |
| PVR+RVOTR+AVR | 0 | 1 |
| PVR+TVP | 0 | 1 |
| PVR+RVOTR+Tricuspid annuloplasty | 1 | 0 |
| PVR+MVR+Tricuspid annuloplasty | 1 | 0 |
| PVR+Coronary fistula (LAD-RV) | 1 | 0 |
| PVR+LV-RA fistula closure | 0 | 1 |
| PVR+RVOTR+Tricuspid annuloplasty+ PAPVR repair | 1 | 0 |
| AVR, aortic valve replacement; LAD, left anterior descending artery; LV, left ventricle; MVR, mitral valve replacement; PAPVR, partial anomalous pulmonary venous return; PVR, pulmonary valve replacement; RA, right atrium; RV, right ventricle; RVOTR, right ventricular outflow tract reconstruction; TVP, tricuspid valve plasty |
Preoperative RV Function
The indices for RV function preoperatively assessed by magnetic resonance imaging were as follows (Table 3). The preoperative RV ejection fraction was significantly lower in patients in Group 1, with a median of 31.3% [24.0–39.8] compared to 44.8% [38.7–56.5] in Group 2 (p=0.028). Similarly, RV end-systolic volume index was significantly greater in Group 1, with no significant differences in other indices including RV end-diastolic volume index. Preoperative magnetic resonance imaging was performed 1.9 months [1.4–1.9] before surgery in Group 1 versus 4.5 months [3.3–5.6] in Group 2.
Table 3 Pre-operative right ventricular function indices assessed by magnetic resonance imaging | Group 1 | Group 2 | p |
|---|
| Number of patients (n) | 4 | 11 | |
| RVEF (%) | 31.3 [24.0–39.8] | 44.8 [38.7–56.5] | 0.028 |
| RVESV (mL) | 209.8 [146.2–274.6] | 107.9 [60.7–144.2] | 0.025 |
| RVESVI (ml/m2) | 110.4 [86.7–145.8] | 69.5 [45.0–95.7] | 0.035 |
| RVEDV (mL) | 313.9 [201.8–420.3] | 216.9 [164.5–298.6] | 0.107 |
| RVEDVI (ml/m2) | 163.9 [120.8–221.1] | 146.8 [102.6–179.2] | 0.187 |
| RVCO (l/min) | 6.5 [4.5–9.5] | 6.1 [4.9–9.7] | 0.495 |
| Pulmonary RF (%) | 64.5 [46.8–82.3] | 58.6 [50.8–67.0] | 0.328 |
| Values are presented as median [interquartile range]. RF, regurgitant fraction; RVCO, right ventricular cardiac output; RVEDV, right ventricular end-diastolic volume; RVEDVI, right ventricular end-diastolic volume index; RVEF, right ventricular ejection fraction; RVESV, right ventricular end-systolic volume; RVESVI, right ventricular end-systolic volume index |
Overall Survival
The median follow-up period after PVR was 1.1years [0.8–1.3] in Group 1 and 3.1 years [2.9–11.0] in Group 2 (p=0.005), respectively. No patient in Group 2 has died, whereas 3 out of the 4 patients died in Group 1; two postoperative in-hospital deaths and one late death after discharge. The in-hospital deaths occurred on postoperative day 367 (due to heart failure), and postoperative day 102 (a combination of heart failure and sepsis). The late death occurred approximately one year after discharge from PVR, its cause being attributed to heart failure, with suspected coronary ischemia as a contributing factor. The remaining one patient in Group 1 was lost scheduled follow-up.
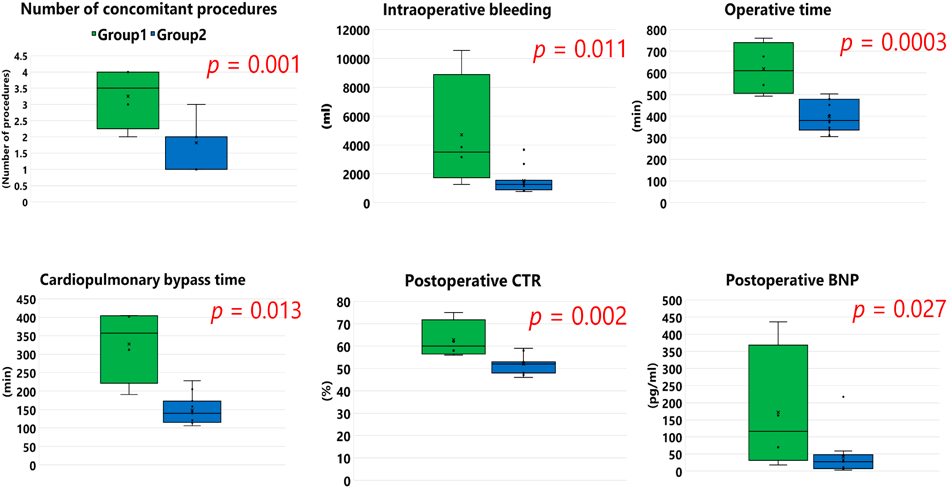
Comparison of Peri- and Post-Operative Factors between the Two Groups (Fig. 1)
A median number of concomitant procedures was 3.5 [2.8–4.0] in Group 1 and 2.0 [1.5–2.0] in Group 2 (p=0.001). The median operative time was 610 minutes [531–697] in Group 1 and 380 minutes [341–466] in Group 2 (p=0.0003). As shown in Fig. 1, other results were also significantly lower in Group 2 such as cardiopulmonary bypass time, amount of blood loss, post-operative cardio-thoracic ratio, and postoperative serum brain natriuretic peptide level. Serum concentration of brain natriuretic peptide was measured at 5.8 months [3.8–10.8] after PVR in Group 1 and 37.6 months [33.7–134.0] in Group 2; chest X-ray was checked at 9.8 months [6.2–13.9] postoperatively in Group 1 and 40.4 months [36.3–131.2] in Group 2.
Post-Operative RV Function (Table 4)
Postoperative magnetic resonance imaging could not be compared due to the small number of investigations performed in Group 1. The median postoperative RV diameter measured by ultrasound echocardiography was 43.3 mm [40.6–45.3] in Group 1 and 38.0 mm [32.0–43.4] in Group 2 (p=0.048). The median tricuspid annular plane systolic excursion and RV fractional area change were significantly higher in Group 2. The timing of postoperative echocardiography was 8.2 months [5.6–12.1] after surgery in Group 1 and 38.3 months [36.3–134.4] in Group 2.
Table 4 Post-operative right ventricular function indices assessed by ultrasound echocardiography | Group 1 | Group 2 | p |
|---|
| Number of Patients (n) | 4 | 11 | |
| TRPG (mmHg) | 35.5 [27.0–46.5] | 29.0 [27.0–33.3] | 0.104 |
| RVD (mm) | 43.3 [40.6–45.3] | 38.0 [32.0–43.4] | 0.048 |
| TAPSE (mm) | 10.1 [9.3–10.4] | 11.6 [10.3–13.9] | 0.024 |
| RVEDA (cm2) | 30.4 [27.1–36.2] | 25.1 [22.1–30.0] | 0.206 |
| RVESA (cm2) | 22.2 [18.3–27.4] | 15.3 [12.9–18.7] | 0.128 |
| RVFAC (%) | 31.0 [25.6–34.0] | 38.0 [37.5–42.5] | 0.022 |
| Values are presented as median [interquartile range]. RVD, right ventricular diameter; RVEDA, right ventricular end-diastolic area; RVESA, right ventricular end-systolic area; RVFAC, right ventricular fractional area change; TAPSE, tricuspid annular plane systolic excursion; TRPG, tricuspid regurgitant pressure gradient |
This study highlighted that interruption of scheduled follow-up by a cardiology specialist in TOF patients repaired using a TAP delayed the timing of PVR, which resulted in poor survival rate. Indeed, the number of concomitant procedures, the amount of intraoperative blood loss, operative time, and cardiopulmonary bypass time were significantly less favorable in Group 1.
Different from municipal hospitals, regional institutions in countryside such as our center often experience patients who had a history of TOF repair during childhood and in whom follow-up was discontinued long time ago because of their relocation or individual life situation mainly. They did not have an opportunity to understand late clinical features of repaired TOF and the importance of regular and close follow-up to detect any signs of right heart failure appropriately. Indeed, median age at PVR in Group 1 was 49 years old, and median duration from TOF repair to PVR was 41 years. The therapeutic effects of PVR after TOF repair are believed to include reduction of right heart failure symptoms and improvement in overall prognosis. Early intervention is considered to enhance effectiveness of the treatment.1–7) Late referral for PVR in repaired TOF patients required combined procedures other than PVR, such as tricuspid valve surgery, anti-arrythmic surgery, and procedures for left heart lesions. The number of concomitant procedures in Group 1 was 1.8 times as much as that in Group 2. Prolonged cardiopulmonary bypass time, challenging chest re-entry, and dissection of tough adhesion around the RVOT/the pulmonary artery required a considerable amount of blood transfusion. The combination of delayed recovery from pre-existing right heart failure and multiple organ dysfunction postoperatively developed led to poor survival outcomes after PVR. To prevent such circumstances in the future, it is essential for cardiologists familiar with congenital heart disease to provide patients and their families clear explanations of long-term issues and prognoses, and to ensure appropriate follow-up care in the outpatient setting. Implantable devices for transcatheter pulmonary valve implantation (TPVI) together with their delivery options are developing for forthcoming treatments in repaired TOF patients.9–12)
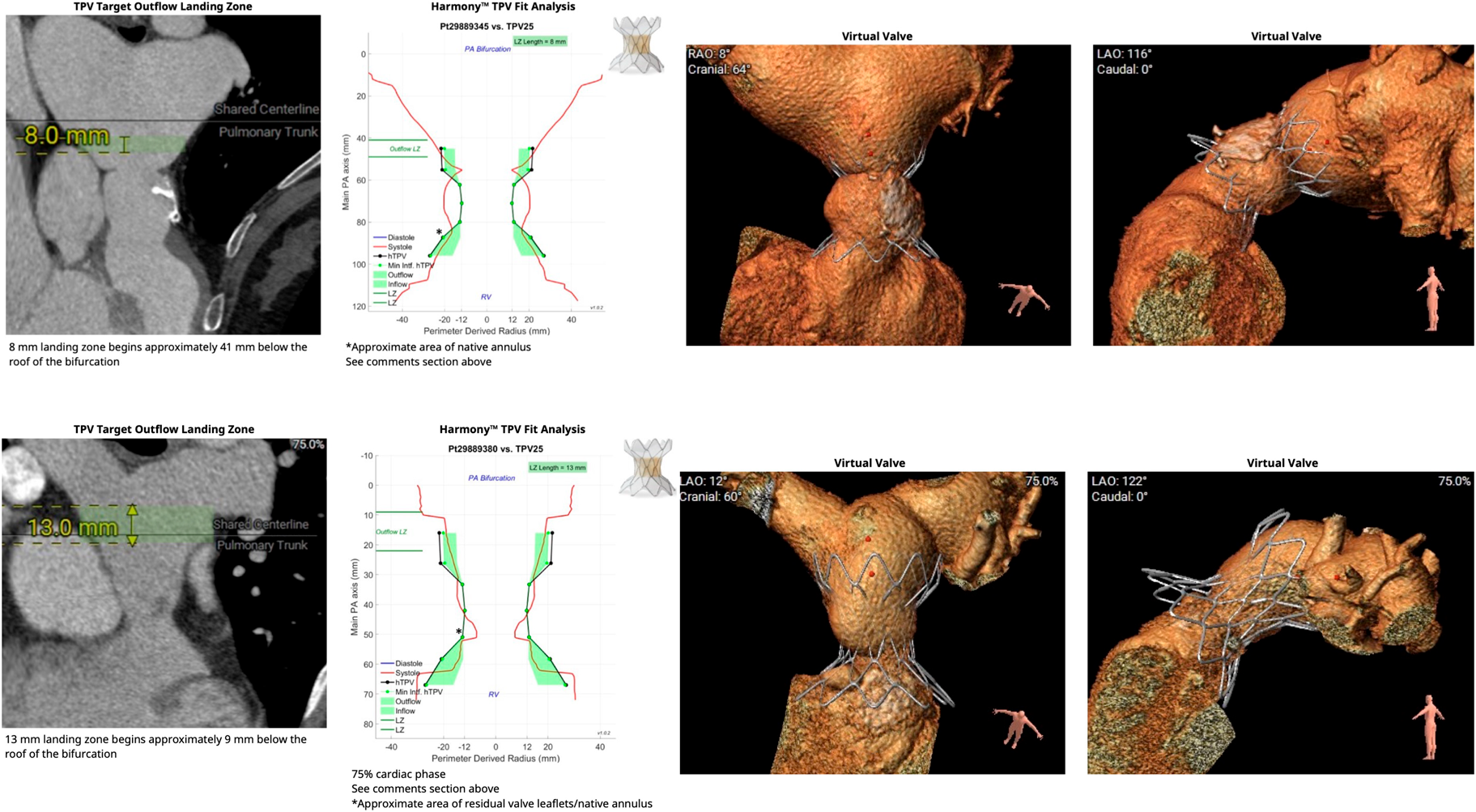
The Harmony valve is a self-expandable device designed to sit within a relatively large native RVOT.13) This device has been available in Japan since March 2023, currently, 22 mm- and 25 mm-sized products can be used. Computational fitting analysis in 2 of Group 1 patients concluded that Harmony valve implantation would have been feasible in either of them (Fig. 2). Such TPVI, which has recently become available in Japan, would offer a less invasive option to reduce pulmonary regurgitation and might lead to improved outcomes in patients with a high risk of mortality due to poor right heart function at the time of PVR as a result of discontinued follow-up. On the other hand, it can be inferred that patients who died in the longer term after discharge were unable to fully recover essential cardiac function despite the fact that they survived the surgical intervention. Prolonged RV overload might not adequately promote recovery of RV function, even if TPVI were technically feasible. The importance of consecutive and appropriate follow-up, therefore, remains unchanged in the TPVI era.
The most significant limitation of this study was its small number of enrolled patients. Especially, the Group 1 consisted of only 4 patients. Secondly, it was not possible to compare pre-operative and post-operative magnetic resonance imaging results due to the minimal opportunity of postoperative investigation. As an additional limitation of this study, a comparison could not be established between the patients in terms of comparable age at surgery or time since previous surgery. Future reports may help better understanding of the importance of follow-up by a cardiologist by comparing patients aged 20-years or 40/50-years who have been followed up by a cardiologist with those who have not.
Repaired Fallot patients who did not receive chronological follow-up by cardiology specialists showed extremely poor prognostic outcomes after surgical PVR. This study shows that it is important for medical professionals to appropriately explain the long-term results and prognosis of TOF and to continue regularly outpatient treatment.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
There are no additional data to disclose.
Author Contribution
HN wrote the manuscript (original draft) and contributed to the creation of the figures and table. TH and TS supervised and authorized this study. AH, TK, YF and YI performed the surgery and/or perioperative patient management. All authors read and approved the manuscript. Of note, the authors declare that the entire manuscript was written by Japanese, then translated and edited using Large Language Models, or ChatGPT (DeepL), then finally edited by native English speaker.
Ethics Approval and Consent for Publication
This study was approved by Saitama Medical University International Medical Center Institutional Review Board (Approval number 2023-077, 12/10/2023).
引用文献References
1) Harrild DM, Berul CI, Cecchin F, et al: Pulmonary valve replacement in tetralogy of Fallot: Impact on survival and ventricular tachycardia. Circulation 2009; 119: 445–451
2) Therrien J, Samuel S, McLaughlin P, et al: Pulmonary valve replacement in adults late after repair of tetralogy of Fallot: Are we operating too late? J Am Coll Cardiol 2000; 36: 1670–1675
3) Romeo JLR, Takkenberg JJM, Cuypers JAAE, et al: Timing of pulmonary valve replacement in patients with corrected Fallot to prevent QRS prolongation. Eur J Cardiothorac Surg 2020; 58: 559–566
4) Gatzoulis MA, Balaji S, Webber SA, et al: Risk factors for arrythmia and sudden cardiac death late after repair of Tetralogy of Fallot: A multicenter study. Lancet 2000; 356: 975–981
5) Lim C, Lee JY, Kim WH, et al: Early replacement of pulmonary valve after repair of tetralogy: Is it really beneficial? Eur J Cardiothorac Surg 2004; 25: 728–734
6) Dorobantu DM, Sharabiani MTA, Taliotis D, et al: Age over 35 years is associated with increased mortality after pulmonary valve replacement in repaired tetralogy of Fallot: Results from the UK National Congenital Heart Disease Audit database. Eur J Cardiothorac Surg 2020; 58: 825–831
7) Therrien J, Provost Y, Merchant N, et al: Optimal timing for pulmonary valve replacement in adults after tetralogy of Fallot repair. Am J Cardiol 2005; 95: 779–782
8) Horose N, Matsuo K, Kabasawa M, et al: The mid-term effects of pulmonary valve replacement on right ventricular function in adult patients with tetralogy of Fallot. Jpn Soc Adult Cong Heart Disease 2013; 2: 22–28
9) Benson LN, Gillespie MJ, Bergersen L, et al: Three-year outcomes from the harmony native outflow tract early feasibility study. Circ Cardiovasc Interv 2020; 13: e008320
10) Law MA, Chatterjee A: Transcatheter pulmonic valve implantation: Techniques, current roles, and future implications. World J Cardiol 2021; 13: 117–129
11) Demkow M, Rużyłło W, Biernacka EK, et al: Percutaneous Edwards SAPIENTM valve implantation for significant pulmonary regurgitation after previous surgical repair with a right ventricular outflow patch. Catheter Cardiovasc Interv 2014; 83: 474–481
12) Levi DS, Sinha S, Salem MM, et al: Transcatheter native pulmonary valve and tricuspid valve replacement with the sapien XT: Initial experience and development of a new delivery platform. Catheter Cardiovasc Interv 2016; 88: 434–443
13) Bergersen L, Benson L, Gillespie MJ, et al: Harmony feasibility trial. JACC Cardiovasc Interv 2017; 10: 1763–1773